
Travere Therapeutics Inc
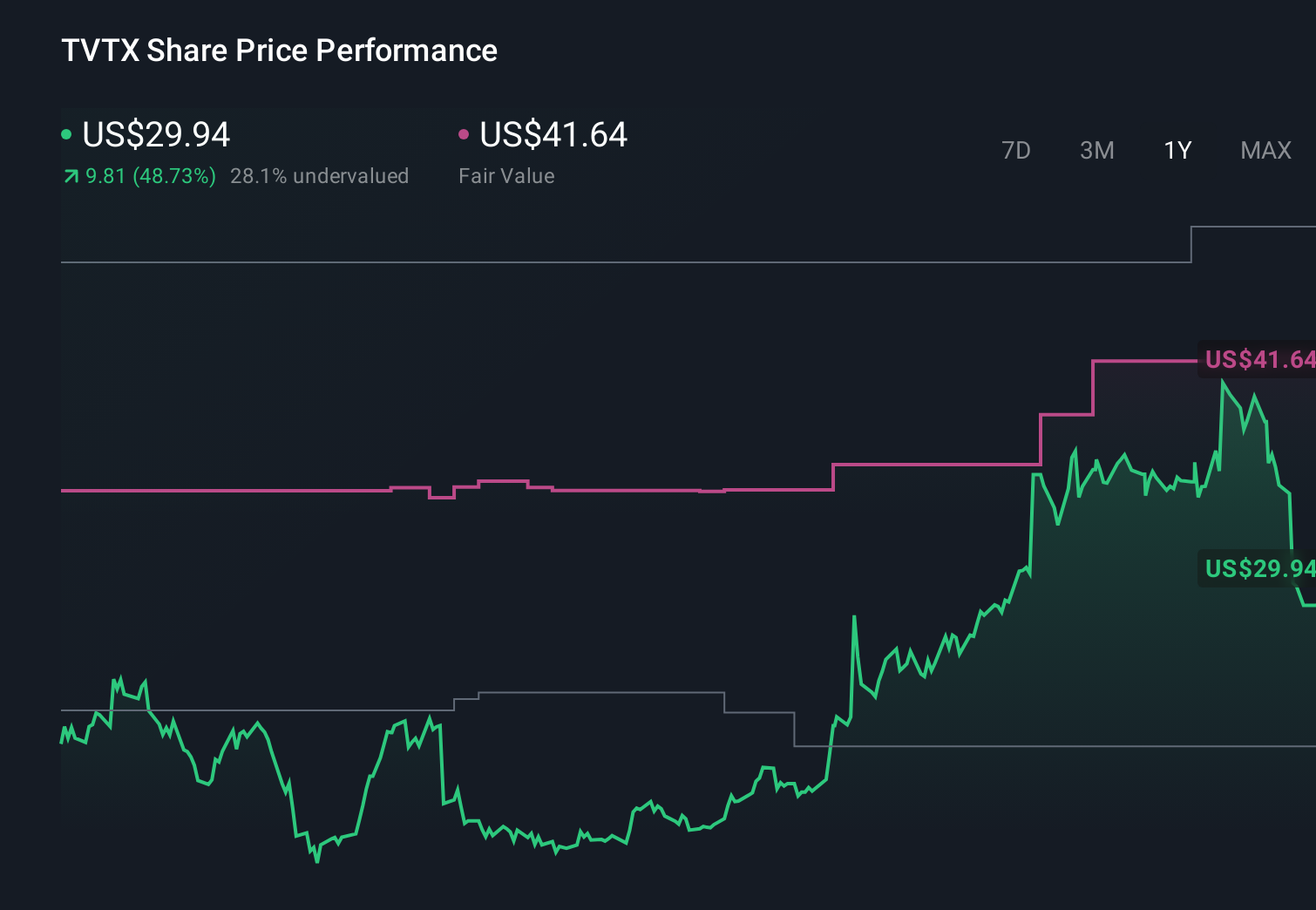
$ 30.88
0.49%
24 Feb - close price
- Market Cap 2,762,905,000 USD
- Current Price $ 30.88
- High / Low $ 31.39 / 30.19
- Stock P/E N/A
- Book Value 1.26
- EPS -1.03
- Next Earning Report -
- Dividend Per Share N/A
- Dividend Yield 0 %
- Next Dividend Date -
- ROA -0.07 %
- ROE -0.58 %
- 52 Week High 42.13
- 52 Week Low 12.91
About
Travere Therapeutics, Inc., a biopharmaceutical company, focuses on the identification, development and commercialization of therapies for the treatment of rare diseases. The company is headquartered in San Diego, California.
Analyst Target Price
$42.71
Quarterly Earnings
| Dec 2025 | Sep 2025 | Jun 2025 | Mar 2025 | Dec 2024 | Sep 2024 | Jun 2024 | Mar 2024 | Dec 2023 | Sep 2023 | Jun 2023 | Mar 2023 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reported Date | 2025-12-31 | 2025-11-05 | 2025-07-30 | 2025-05-05 | 2025-02-13 | 2024-10-31 | 2024-08-01 | 2024-05-06 | 2024-03-12 | 2023-11-07 | 2023-08-03 | 2023-05-04 |
| Reported EPS | 0.37 | 0.59 | 0.13 | -0.19 | -0.47 | -0.7 | -0.91 | -1.76 | -1.18 | -1.17 | -1.13 | -1.27 |
| Estimated EPS | 0.3729 | -0.287 | -0.2715 | -0.3943 | -0.3786 | -0.68 | -0.86 | -0.99 | -1.27 | -0.14 | -1.16 | -1.17 |
| Surprise | -0.0029 | 0.877 | 0.4015 | 0.2043 | -0.0914 | -0.02 | -0.05 | -0.77 | 0.09 | -1.03 | 0.03 | -0.1 |
| Surprise Percentage | -0.7777% | 305.5749% | 147.8821% | 51.8133% | -24.1416% | -2.9412% | -5.814% | -77.7778% | 7.0866% | -735.7143% | 2.5862% | -8.547% |
Next Quarterly Earnings
| Reported Date |
| Fiscal Date Ending |
| Estimated EPS |
| Currency |
Next Dividend Records
| Dividend per share (year): | - |
| Dividend Yield | - |
| Next Dividend Date | - |
| Ex-Dividend Date | - |
Recent News: TVTX
2026-02-25 04:53:29
Travere Therapeutics reported a significant revenue surge to US$490.73 million in 2025, up from US$233.18 million in 2024, and narrowed its net loss to US$25.55 million, driven by the strong uptake of FILSPARI. This financial improvement reinforces confidence in the company's flexibility ahead of a key FDA decision in April 2026 for FILSPARI's sNDA in FSGS. While the strong performance validates the company's financial strategy, investors are cautioned about potential regulatory and pricing pressures.
2026-02-24 12:53:29
Travere Therapeutics (TVTX) reported significant growth for its drug Filspari in 2025, with sales reaching $322 million, a 144% increase year-over-year. This growth was driven by expanded physician confidence, favorable reimbursement, and updated clinical guidelines. The company is actively pursuing the expansion of Filspari into FSGS and resuming the Phase 3 Harmony study for Pegtobatinase, while expanding its field team to support these initiatives.
2026-02-23 10:31:59
Travere Therapeutics (TVTX) recently reported strong quarterly earnings, exceeding market expectations with increased revenue and a narrower loss due to higher physician adoption and pipeline advancements. Despite a year-to-date share price decline of 23.10%, recent sentiment has improved, with the stock seen as 28.3% undervalued by some narratives, suggesting a fair value of $42.36 against its current trading price of $30.39. This valuation is contingent on the continued success of its key pipeline drugs and controlled R&D and SG&A spending.
2026-02-23 06:53:29
Travere Therapeutics (TVTX) has drawn attention after exceeding market expectations with its quarterly earnings, stronger revenue, and narrowed losses, fueled by increased physician adoption and pipeline advancements. Despite a recent share price rebound, the stock is still down 23.10% year-to-date, trading at a discount to analyst targets. The company's valuation narrative suggests it is 28.3% undervalued at $42.36, provided its key drug FILSPARI continues on track and R&D/SG&A spending is managed effectively.
2026-02-21 11:40:14
Bank of America Securities analyst Jason Zemansky reiterated a Buy rating for Travere Therapeutics (TVTX) with a $44.00 price target, based on the company's recent strong financial performance. This positive outlook is supported by a similar Buy rating from H.C. Wainwright, although another analyst maintained a Hold rating. The company reported significant revenue growth and a net profit in its latest quarterly earnings.

2026-02-21 03:52:52
The FDA has granted accelerated approval to Filspari (sparsentan) for reducing proteinuria in adults with primary IgA nephropathy (IgAN) at risk of rapid disease progression. This approval, based on clinical trial data showing a significant reduction in proteinuria compared to irbesartan, positions Filspari as a new treatment option for IgAN patients. The PROTECT study will continue to assess its effect on kidney function decline, with results expected in late 2023.


