
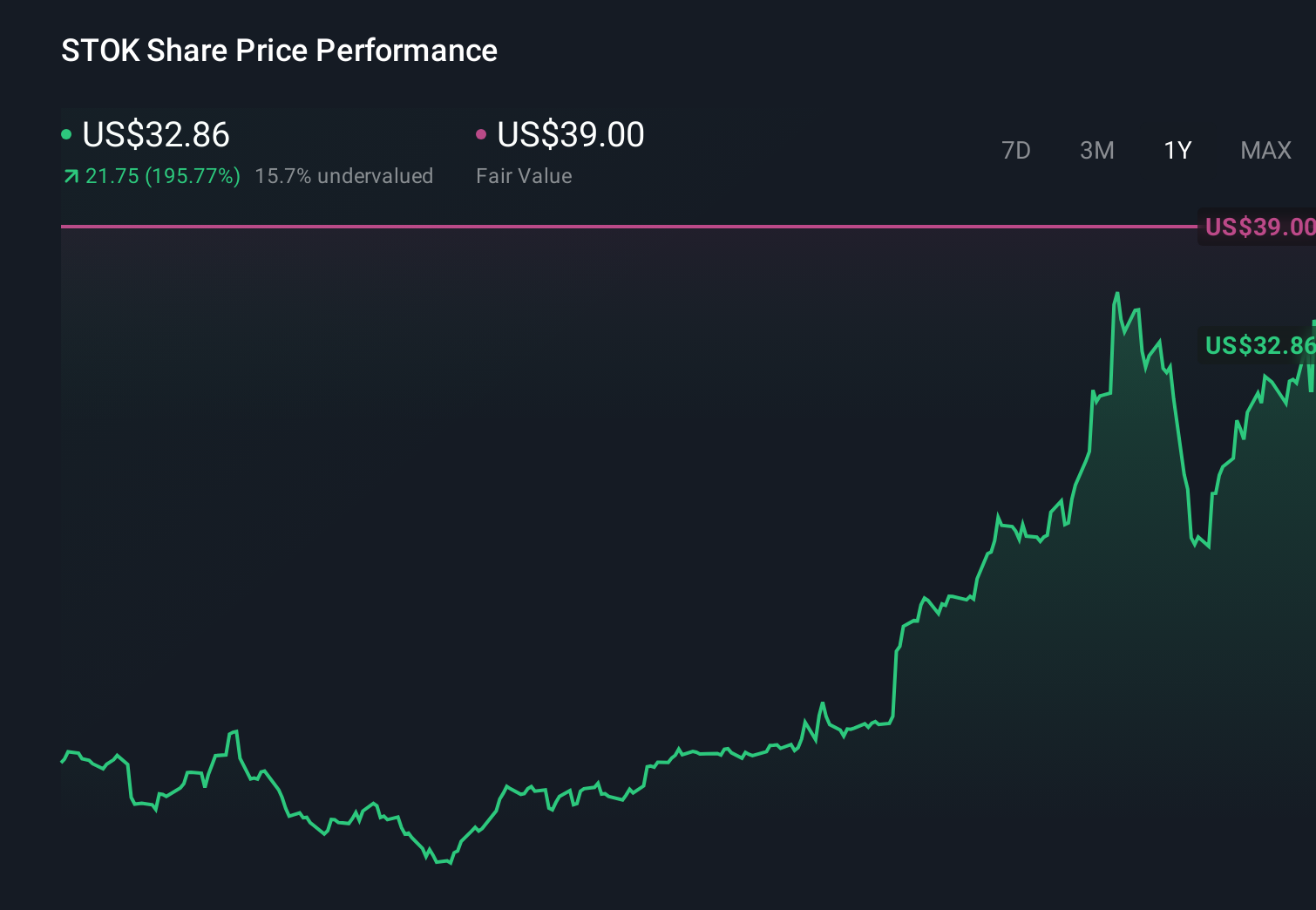
Stoke Therapeutics Inc
$ 35.50
-2.01%
24 Feb - close price
- Market Cap 2,069,354,000 USD
- Current Price $ 35.50
- High / Low $ 37.05 / 35.30
- Stock P/E 53.28
- Book Value 5.59
- EPS 0.68
- Next Earning Report 2026-03-17
- Dividend Per Share N/A
- Dividend Yield 0 %
- Next Dividend Date -
- ROA 0.05 %
- ROE 0.15 %
- 52 Week High 38.69
- 52 Week Low 5.35
About
Stoke Therapeutics, Inc., an early-stage biopharmaceutical company, develops new antisense oligonucleotide (ASO) drugs to treat the underlying causes of serious genetic diseases in the United States. The company is headquartered in Bedford, Massachusetts.
Analyst Target Price
$41.89
Quarterly Earnings
| Sep 2025 | Jun 2025 | Mar 2025 | Dec 2024 | Sep 2024 | Jun 2024 | Mar 2024 | Dec 2023 | Sep 2023 | Jun 2023 | Mar 2023 | Dec 2022 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reported Date | 2025-11-04 | 2025-08-12 | 2025-05-05 | 2025-03-24 | 2024-11-05 | 2024-08-07 | 2024-05-06 | 2024-03-25 | 2023-11-07 | 2023-08-07 | 2023-05-04 | 2023-03-06 |
| Reported EPS | -0.65 | -0.4 | 1.9 | -0.18 | -0.4691 | -0.46 | -0.57 | -0.6 | -0.55 | -0.69 | -0.53 | -0.65 |
| Estimated EPS | -0.58 | -0.5 | -0.0905 | -0.5302 | -0.53 | -0.56 | -0.61 | -0.63 | -0.63 | -0.64 | -0.7 | -0.7 |
| Surprise | -0.07 | 0.1 | 1.9905 | 0.3502 | 0.0609 | 0.1 | 0.04 | 0.03 | 0.08 | -0.05 | 0.17 | 0.05 |
| Surprise Percentage | -12.069% | 20% | 2199.4475% | 66.0505% | 11.4906% | 17.8571% | 6.5574% | 4.7619% | 12.6984% | -7.8125% | 24.2857% | 7.1429% |
Next Quarterly Earnings
| Dec 2025 | |
|---|---|
| Reported Date | 2026-03-17 |
| Fiscal Date Ending | 2025-12-31 |
| Estimated EPS | -0.75 |
| Currency | USD |
Next Dividend Records
| Dividend per share (year): | - |
| Dividend Yield | - |
| Next Dividend Date | - |
| Ex-Dividend Date | - |
Recent News: STOK
2026-02-24 05:25:29
Stoke Therapeutics announced a 13.5% stock increase after dosing the first patient in its STK-002 OSPREY trial for Autosomal Dominant Optic Atrophy. This milestone expands its TANGO platform into ophthalmology, creating a second potential rare disease franchise beyond its ongoing Dravet syndrome trial. While important scientifically, the near-term focus remains on the Phase 3 Dravet trial data readout planned for mid-2027.
2026-02-23 12:58:39
Stoke Therapeutics is nearing a critical period as its stock approaches a 52-week high, driven by anticipation for key clinical trial data. The company's lead candidate, zorevunersen, is in Phase 3 for Dravet Syndrome, with updates on patient recruitment and regulatory discussions being closely watched. Additionally, the early-stage STK-002 for Autosomal Dominant Optic Atrophy is under scrutiny for its safety profile and dose-escalation, making clinical execution paramount for the biotech's market valuation.
2026-02-20 21:39:31
Stoke Therapeutics, Inc. (STOK) shows promising growth with a market capitalization of $1.96 billion and a robust revenue increase of 117.20%. Analysts are bullish, projecting an average target price of $41.89, indicating a potential upside of 21.88%. The company's innovative TANGO platform is advancing treatments for genetic diseases, with lead candidates STK-002 and Zorevunersen (STK-001) in various stages of clinical development.

2026-02-18 21:28:17
Stoke Therapeutics, Inc. announced that it granted stock options to five new employees to purchase an aggregate of 22,950 shares of common stock. These grants, effective February 17, 2026, serve as a material inducement for their employment under Nasdaq Listing Rule 5635(c)(4). The options have an exercise price of $31.09 per share, consistent with the closing price on the grant date, and vest over a four-year period.

2026-02-18 01:57:25
This article provides an AI-driven liquidity mapping and sentiment analysis for Stoke Therapeutics Inc. (NASDAQ: STOK), characterizing its near-term sentiment as neutral amid mid and long-term strength. It outlines three distinct trading strategies—Position, Momentum Breakout, and Risk Hedging—with specific entry, target, and stop-loss levels. The analysis also highlights key support and resistance levels across different time horizons, alongside a notable risk-reward short setup.

2026-02-15 16:58:02
Stoke Therapeutics highlighted long-term, durable seizure reductions and cognitive improvements in its Dravet syndrome program, with Phase 3 enrollment expected to conclude in Q2 2026 and pivotal data by mid-2027. The company also announced the initiation of a new Phase I/II program for autosomal dominant optic atrophy (ADOA), with first patient dosed. Stoke reported a strong financial position, ending 2025 with $400 million in cash, providing runway into 2028.


