
Penumbra Inc
$ 318.74
-0.07%
02 Jun - close price
- Market Cap 12,544,758,000 USD
- Current Price $ 318.74
- High / Low $ 319.45 / 318.12
- Stock P/E 73.49
- Book Value 36.39
- EPS 4.34
- Next Earning Report 2026-07-28
- Dividend Per Share N/A
- Dividend Yield 0 %
- Next Dividend Date -
- ROA 0.07 %
- ROE 0.13 %
- 52 Week High 362.41
- 52 Week Low 221.26
About
Penumbra, Inc. designs, develops, manufactures, and markets medical devices in the United States, Europe, Canada, Australia, and internationally. The company is headquartered in Alameda, California.
Analyst Target Price
$363.36
Quarterly Earnings
| Mar 2026 | Dec 2025 | Sep 2025 | Jun 2025 | Mar 2025 | Dec 2024 | Sep 2024 | Jun 2024 | Mar 2024 | Dec 2023 | Sep 2023 | Jun 2023 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reported Date | 2026-05-06 | 2026-02-25 | 2025-11-05 | 2025-07-29 | 2025-04-23 | 2025-02-18 | 2024-10-30 | 2024-07-30 | 2024-05-07 | 2024-02-22 | 2023-11-02 | 2023-08-01 |
| Reported EPS | 0.82 | 1.18 | 0.97 | 0.86 | 0.83 | 0.97 | 0.85 | 0.64 | 0.41 | 0.76 | 0.67 | 0.43 |
| Estimated EPS | 1.08 | 1.1 | 0.92 | 0.82 | 0.67 | 0.8937 | 0.69 | 0.56 | 0.38 | 0.7 | 0.45 | 0.28 |
| Surprise | -0.26 | 0.08 | 0.05 | 0.04 | 0.16 | 0.0763 | 0.16 | 0.08 | 0.03 | 0.06 | 0.22 | 0.15 |
| Surprise Percentage | -24.0741% | 7.2727% | 5.4348% | 4.878% | 23.8806% | 8.5375% | 23.1884% | 14.2857% | 7.8947% | 8.5714% | 48.8889% | 53.5714% |
Next Quarterly Earnings
| Jun 2026 | |
|---|---|
| Reported Date | 2026-07-28 |
| Fiscal Date Ending | 2026-06-30 |
| Estimated EPS | 1.14 |
| Currency | USD |
Next Dividend Records
| Dividend per share (year): | - |
| Dividend Yield | - |
| Next Dividend Date | - |
| Ex-Dividend Date | - |
Recent News: PEN
2026-06-01 09:09:13
Penumbra, Inc. (PEN) is highlighted as an intriguing investment opportunity in the medical devices industry, with a market capitalization of $12.52 billion and robust revenue growth of 15.60%. The company specializes in innovative medical devices like the Indigo System for thrombectomy, backed by strong free cash flow and a respectable return on equity. Analyst consensus suggests a generally positive outlook with an average target price indicating a potential upside of approximately 14.16%.

2026-05-31 03:09:18
Faruqi & Faruqi, LLP is investigating potential claims against Penumbra, Inc. (NYSE:PEN) and reminds investors of the March 16, 2021 deadline to seek the role of lead plaintiff in a federal securities class action. The lawsuit alleges that Penumbra made false and misleading statements regarding its Jet 7 Xtra Flex catheter, which had known design defects and safety issues leading to serious injuries and deaths. The firm encourages investors who suffered losses exceeding $100,000 to contact them to discuss their legal rights.

2026-05-30 07:39:39
Faruqi & Faruqi, LLP is investigating potential securities claims against Penumbra, Inc. (NYSE:PEN) and reminds investors of the March 16, 2021 deadline to seek the role of lead plaintiff in a federal securities class action. The lawsuit alleges that Penumbra made false and misleading statements about the safety of its Jet 7 Xtra Flex catheter, which was later recalled due to design defects causing injury or death. Investors who suffered losses exceeding $100,000 between August 3, 2020, and December 15, 2020, are encouraged to contact the firm.
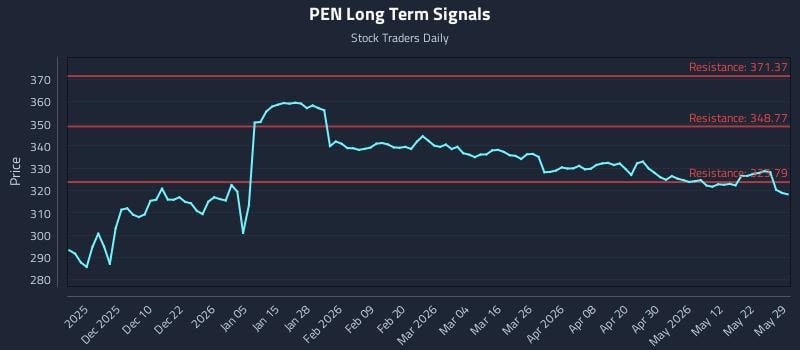
2026-05-30 04:38:48
This article provides an AI-driven analysis for Penumbra Inc. (PEN), highlighting weak near-term sentiment despite potential long-term strength. It offers three distinct trading strategies—Position Trading, Momentum Breakout, and Risk Hedging—along with multi-timeframe signal analysis indicating support and resistance levels. The analysis suggests choppy conditions due to divergent sentiment across different time horizons.

2026-05-29 08:09:15
Penumbra Inc. shares have stabilized around $320 after a recent decline, valuing the medical device company at approximately $13 billion. This pullback has shifted investors' focus to the company's valuation metrics, profitability, and product pipeline. While trading at a premium compared to broader healthcare companies, Penumbra's future performance will depend on sustained revenue growth, operating efficiency, and new product adoption in its neurovascular and thrombectomy sectors.

2026-05-28 10:39:23
This article from Penumbra, Inc. emphasizes the critical importance of early stroke recognition and treatment, highlighting that "time is brain" due to rapid brain cell death. It details stroke prevalence and risk factors, introduces the B.E. F.A.S.T acronym for symptom identification, and discusses the value of knowing certified stroke centers and recent advancements like mechanical thrombectomy for improved patient outcomes.


