
Enanta Pharmaceuticals Inc
$ 11.28
-3.84%
05 Jun - close price
- Market Cap 326,841,000 USD
- Current Price $ 11.28
- High / Low $ 11.68 / 11.25
- Stock P/E N/A
- Book Value 4.01
- EPS -2.60
- Next Earning Report -
- Dividend Per Share N/A
- Dividend Yield 0 %
- Next Dividend Date -
- ROA -0.12 %
- ROE -0.59 %
- 52 Week High 17.15
- 52 Week Low 6.43
About
Enanta Pharmaceuticals, Inc., a biotechnology company, discovers and develops small molecule drugs for the treatment of viral infections and liver diseases. The company is headquartered in Watertown, Massachusetts.
Analyst Target Price
$20.14
Quarterly Earnings
| Mar 2026 | Dec 2025 | Sep 2025 | Jun 2025 | Mar 2025 | Dec 2024 | Sep 2024 | Jun 2024 | Mar 2024 | Dec 2023 | Sep 2023 | Jun 2023 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reported Date | 2026-05-11 | 2026-02-09 | 2025-11-17 | 2025-08-11 | 2025-05-12 | 2025-02-10 | 2024-11-25 | 2024-08-05 | 2024-05-06 | 2024-02-07 | 2023-11-20 | 2023-08-07 |
| Reported EPS | -0.4508 | -0.42 | -0.87 | -0.85 | -1.06 | -1.05 | -1.36 | -1.07 | -1.47 | -1.58 | -1.33 | -1.86 |
| Estimated EPS | -0.7836 | -0.81 | -1.01 | -0.88 | -1.14 | -1.35 | -0.92 | -1.45 | -1.33 | -1.19 | -2.15 | -2.34 |
| Surprise | 0.3328 | 0.39 | 0.14 | 0.03 | 0.08 | 0.3 | -0.44 | 0.38 | -0.14 | -0.39 | 0.82 | 0.48 |
| Surprise Percentage | 42.4706% | 48.1481% | 13.8614% | 3.4091% | 7.0175% | 22.2222% | -47.8261% | 26.2069% | -10.5263% | -32.7731% | 38.1395% | 20.5128% |
Next Quarterly Earnings
| Reported Date |
| Fiscal Date Ending |
| Estimated EPS |
| Currency |
Next Dividend Records
| Dividend per share (year): | - |
| Dividend Yield | - |
| Next Dividend Date | - |
| Ex-Dividend Date | - |
Recent News: ENTA
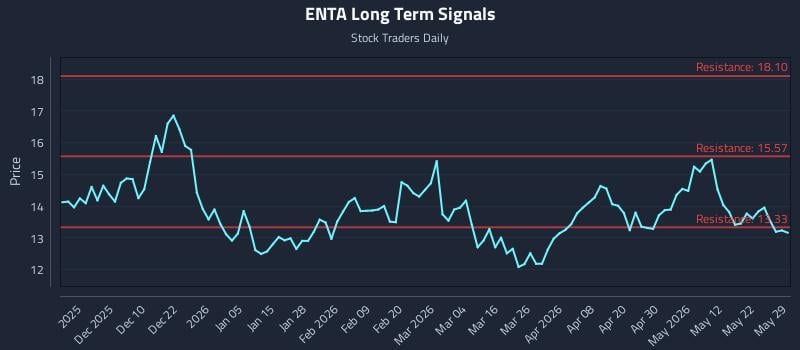
2026-05-30 19:58:00
Enanta Pharmaceuticals Inc. (NASDAQ: ENTA) is showing weak near and mid-term sentiment, challenging a previously strong long-term outlook. Despite a lack of clear price positioning signals and elevated downside risk, AI models have generated distinct trading strategies including long, breakout, and short positions based on different risk profiles and time horizons. The analysis emphasizes real-time signals, risk management, and multi-timeframe insights for investors.

2026-05-28 13:09:47
Enanta Pharmaceuticals announced that its President and CEO, Jay R. Luly, Ph.D., and Chief Product Strategy Officer, Tara L. Kieffer, Ph.D., will participate in a fireside chat at the Jefferies Global Healthcare Conference on June 4, 2026. The clinical-stage biotechnology company, focused on small molecule drugs for viral infections and immunological diseases, will provide a live webcast of the event on its investor relations website. A replay will also be available for at least 30 days following the presentation.

2026-05-28 10:40:25
Enanta Pharmaceuticals, Inc. announced that its President and CEO, Jay R. Luly, and Chief Product Strategy Officer, Tara L. Kieffer, will participate in a fireside chat at the Jefferies Global Healthcare Conference. The event is scheduled for June 4, 2026, at 11:40 a.m. ET in New York, NY, and a live webcast will be available on Enanta's website. The company focuses on developing small molecule drugs for viral infections and immunological diseases, with ongoing programs in RSV and inflammatory diseases.
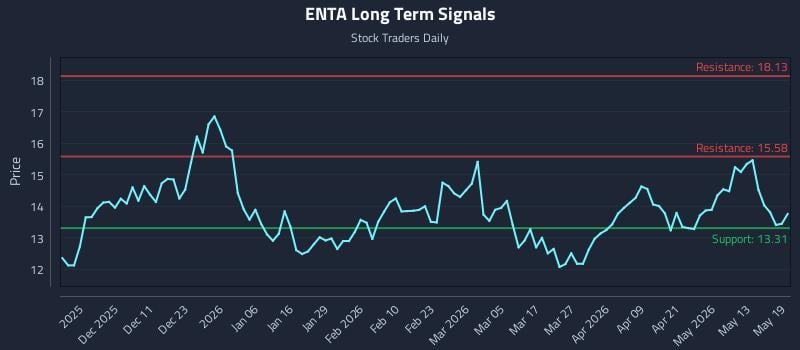
2026-05-19 18:50:00
The article discusses the investment opportunity in Enanta Pharmaceuticals Inc. (NASDAQ: ENTA), highlighting a mid-channel oscillation pattern and a significant 56.7:1 risk-reward setup targeting a 17.1% gain. It provides three distinct AI-generated trading strategies (Position, Momentum Breakout, Risk Hedging) tailored for different risk profiles, along with multi-timeframe signal analysis indicating weak near-term sentiment but strong long-term potential.

2026-05-14 16:39:30
Enanta Pharmaceuticals (ENTA) reported improved Q2 2026 results, with a narrowed basic EPS loss of US$0.45 and revenue of US$17.2 million. The company's trailing twelve-month net loss decreased by US$9.6 million, and quarterly losses have been consistently smaller compared to previous periods. Despite ongoing unprofitability and shareholder dilution, the stock trades at a lower P/S ratio than the industry average, with analysts expecting 17.6% annual revenue growth.
2026-05-13 19:39:49
Enanta Pharmaceuticals (NASDAQ: ENTA) released Q2 results, with revenues meeting analyst predictions at US$17m and statutory losses being 7.7% smaller than expected at US$0.45 per share. Analysts forecast revenues of US$66.9m for 2026, a 3.3% decline, and increased per-share losses to US$2.21, reflecting mixed sentiment. Despite these adjustments, the consensus price target remains US$20.14, indicating that the business is performing largely as expected.


