
Castle Biosciences Inc
$ 30.83
2.97%
25 Feb - close price
- Market Cap 873,908,000 USD
- Current Price $ 30.83
- High / Low $ 31.11 / 29.92
- Stock P/E N/A
- Book Value 16.02
- EPS -0.44
- Next Earning Report 2026-02-26
- Dividend Per Share N/A
- Dividend Yield 0 %
- Next Dividend Date -
- ROA -0.02 %
- ROE -0.03 %
- 52 Week High 44.28
- 52 Week Low 14.59
About
Castle Biosciences Inc. is a leader in the commercial-stage diagnostics sector, specializing in innovative gene-expression profiling tests aimed at improving patient outcomes in skin cancer and related conditions. Its flagship product, DecisionDx®-Melanoma, provides critical insights for risk stratification and treatment pathways in melanoma patients by assessing gene expression levels. The company is dedicated to advancing precision medicine through its proprietary technology platform, which supports the development of additional assays designed to meet significant clinical needs. With a robust pipeline backed by a strong focus on research and development, Castle Biosciences is strategically positioned for growth and aims to enhance both clinical outcomes and shareholder value in the evolving oncology landscape.
Analyst Target Price
$48.12
Quarterly Earnings
| Sep 2025 | Jun 2025 | Mar 2025 | Dec 2024 | Sep 2024 | Jun 2024 | Mar 2024 | Dec 2023 | Sep 2023 | Jun 2023 | Mar 2023 | Dec 2022 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reported Date | 2025-11-03 | 2025-08-04 | 2025-04-30 | 2025-02-26 | 2024-11-04 | 2024-08-05 | 2024-05-02 | 2024-02-28 | 2023-11-02 | 2023-08-02 | 2023-05-03 | 2023-02-28 |
| Reported EPS | -0.02 | 0.15 | -0.9035 | 0.32 | 0.08 | 0.31 | -0.09 | -0.1 | -0.26 | -0.7 | -1.1 | -0.78 |
| Estimated EPS | -0.52 | -0.51 | -0.0657 | 0.04 | -0.06 | -0.27 | -0.33 | -0.54 | -0.76 | -0.91 | -0.89 | -0.84 |
| Surprise | 0.5 | 0.66 | -0.8378 | 0.28 | 0.14 | 0.58 | 0.24 | 0.44 | 0.5 | 0.21 | -0.21 | 0.06 |
| Surprise Percentage | 96.1538% | 129.4118% | -1275.1903% | 700% | 233.3333% | 214.8148% | 72.7273% | 81.4815% | 65.7895% | 23.0769% | -23.5955% | 7.1429% |
Next Quarterly Earnings
| Dec 2025 | |
|---|---|
| Reported Date | 2026-02-26 |
| Fiscal Date Ending | 2025-12-31 |
| Estimated EPS | -0.22 |
| Currency | USD |
Next Dividend Records
| Dividend per share (year): | - |
| Dividend Yield | - |
| Next Dividend Date | - |
| Ex-Dividend Date | - |
Recent News: CSTL

2026-02-25 20:51:51
Castle Biosciences, Inc. experienced a significant stock decline of 7.66% to an intraday low of $27.80 after reporting a disappointing financial quarter ending March 2025. The company's pre-tax profits fell by 116.84% and net profits by 62.58%, accompanied by a concerning debt-equity ratio of -52.39%, raising questions about its financial stability. Despite an 11.70% return over the past year, profits have plummeted by 215.5%, indicating a challenging outlook for the biotechnology firm.

2026-02-25 19:51:51
Castle Biosciences (NASDAQ: CSTL) is scheduled to release its quarterly earnings report. Analysts expect an EPS of $-0.24, and investors will be looking for guidance for the next quarter. The company's stock has shown positive performance, up 20.98% over the last 52 weeks, but it faces challenges regarding market capitalization, revenue decline, and profitability metrics.
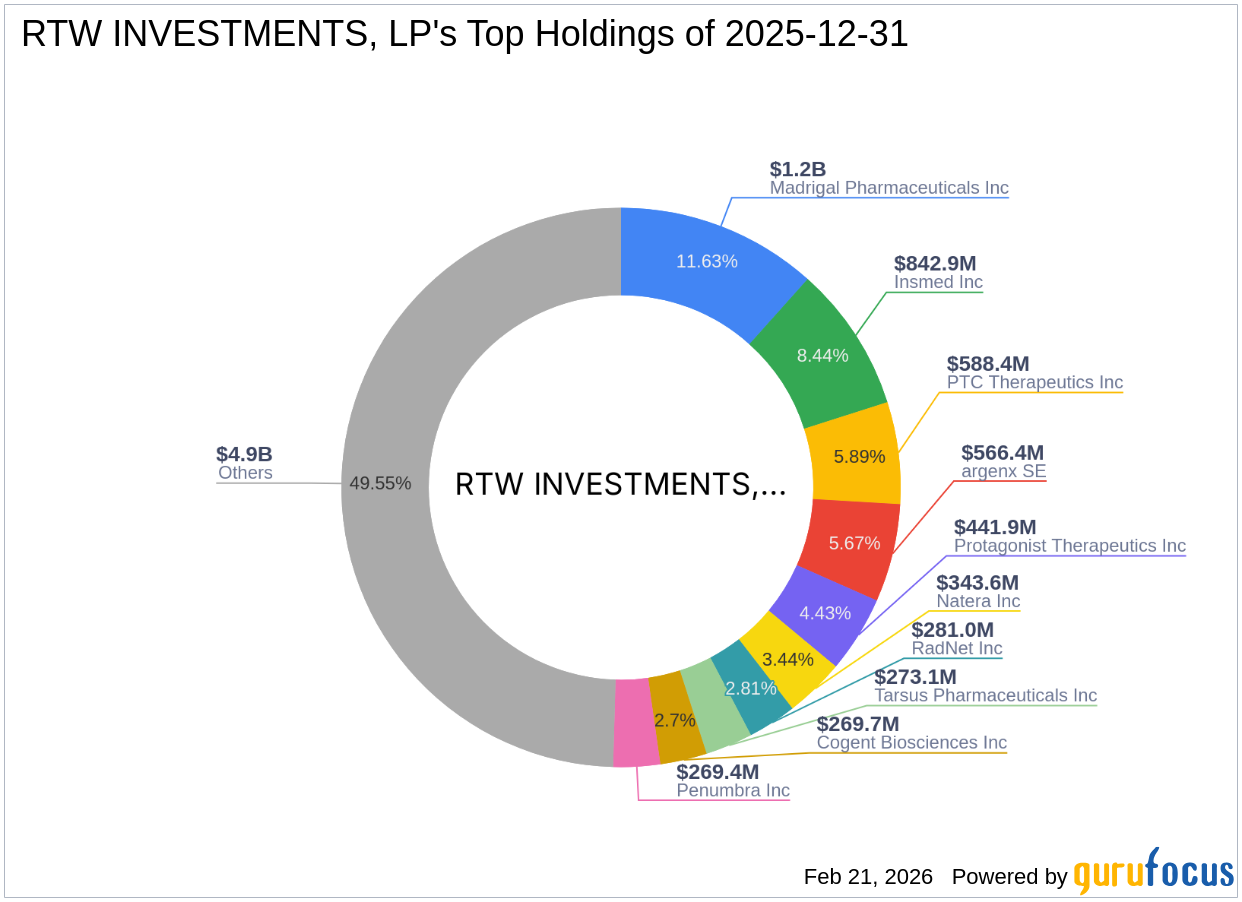
2026-02-21 02:33:19
RTW INVESTMENTS, LP has acquired a substantial stake of 2,115,237 shares in Castle Biosciences Inc. at $38.90 per share, making it 7.20% of their holdings. This strategic investment in the molecular diagnostics company reflects RTW's focus on the healthcare sector and confidence in Castle Biosciences' growth potential. Despite a recent stock price decrease, the company's innovation in medical diagnostics and solid financial strength are highlighted.

2026-02-20 18:05:00
Castle Biosciences announced the publication of a prospective study in the Journal of the American Academy of Dermatology, validating its AdvanceAD-Tx test. The study demonstrates that AdvanceAD-Tx can identify patients with moderate-to-severe atopic dermatitis more likely to respond significantly better and faster to JAK inhibitor therapy compared to Th2-targeted therapies. This non-invasive gene expression profile test aims to guide systemic treatment decisions by aligning therapy choices with a patient's individual disease biology, potentially leading to improved outcomes and streamlined care.

2026-02-20 14:09:01
A prospective study published in the Journal of the American Academy of Dermatology (JAAD) validated Castle Biosciences' AdvanceAD-Tx™ test for identifying patients with moderate-to-severe atopic dermatitis (AD) who are more likely to respond faster and better to JAK inhibitor therapy. The test stratifies patients by molecular profile, helping clinicians align systemic therapy choices with individual disease biology. Patients with a JAK Inhibitor Responder Profile treated with JAKi showed significantly greater improvements in skin clearance, itch reduction, and quality of life compared to those on Th2-targeted therapies.

2026-02-20 11:27:24
Castle Biosciences announced that its AdvanceAD-Tx test can identify patients with moderate-to-severe atopic dermatitis who are more likely to respond significantly better and faster to JAK inhibitor therapy. A prospective study published in JAAD demonstrated that about 30% of patients had a "JAK Inhibitor Responder Profile," achieving EASI-90 nearly four times faster with JAKi treatment compared to Th2-targeted therapies. This molecular insight aims to help clinicians personalize treatment earlier, reducing unnecessary therapy changes and accelerating positive clinical outcomes.


