
Cumberland Pharmaceuticals Inc
$ 4.10
4.59%
25 Feb - close price
- Market Cap 61,336,000 USD
- Current Price $ 4.10
- High / Low $ 4.11 / 3.93
- Stock P/E N/A
- Book Value 1.75
- EPS -0.24
- Next Earning Report 2026-03-03
- Dividend Per Share N/A
- Dividend Yield 0 %
- Next Dividend Date -
- ROA -0.03 %
- ROE -0.13 %
- 52 Week High 7.25
- 52 Week Low 1.85
About
Cumberland Pharmaceuticals Inc., a specialty pharmaceutical company, focuses on the acquisition, development, and marketing of prescription products for hospital acute care, gastroenterology, and rheumatology in the United States and internationally. The company is headquartered in Nashville, Tennessee.
Analyst Target Price
$8.50
Quarterly Earnings
| Sep 2025 | Jun 2025 | Mar 2025 | Dec 2024 | Sep 2024 | Jun 2024 | Mar 2024 | Dec 2023 | Sep 2023 | Jun 2023 | Mar 2023 | Dec 2022 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reported Date | 2025-11-04 | 2025-08-08 | 2025-05-05 | 2025-03-03 | 2024-11-07 | 2024-08-06 | 2024-05-07 | 2024-03-05 | 2023-11-07 | 2023-08-08 | 2023-05-09 | 2023-03-07 |
| Reported EPS | -0.1297 | -0.05 | 0.0824 | -0.1363 | -0.1099 | -0.0769 | -0.1381 | -0.4441 | 0.02 | 0.16 | 0.11 | -0.05 |
| Estimated EPS | 0 | 0 | None | None | None | None | None | None | None | None | None | None |
| Surprise | -0.1297 | -0.05 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Surprise Percentage | None% | None% | None% | None% | None% | None% | None% | None% | None% | None% | None% | None% |
Next Quarterly Earnings
| Dec 2025 | |
|---|---|
| Reported Date | 2026-03-03 |
| Fiscal Date Ending | 2025-12-31 |
| Estimated EPS | |
| Currency | USD |
Next Dividend Records
| Dividend per share (year): | - |
| Dividend Yield | - |
| Next Dividend Date | - |
| Ex-Dividend Date | - |
Recent News: CPIX

2026-02-21 07:20:24
Cumberland Pharmaceuticals is under increased investor scrutiny due to its clinical pipeline, particularly for Ifetroban which recently received FDA Fast Track designation for Duchenne Muscular Dystrophy. While the company's established commercial products provide revenue, investors are closely watching clinical trial data and the sustainability of recent stock performance, especially after a technical breakout. Upcoming quarterly reports and further clinical updates are viewed as key catalysts for future valuation.

2026-02-17 04:58:59
Cumberland Pharmaceuticals (NASDAQ:CPIX) stock recently crossed above its 200-day moving average, trading as high as $3.95 on Monday. Despite this upward movement, analyst sentiment remains largely negative, with a consensus "Sell" rating, although Wall Street Zen recently upgraded the stock to "Hold." The company is a small-cap specialty pharmaceutical firm with a negative P/E ratio and approximately 15.5% institutional ownership.
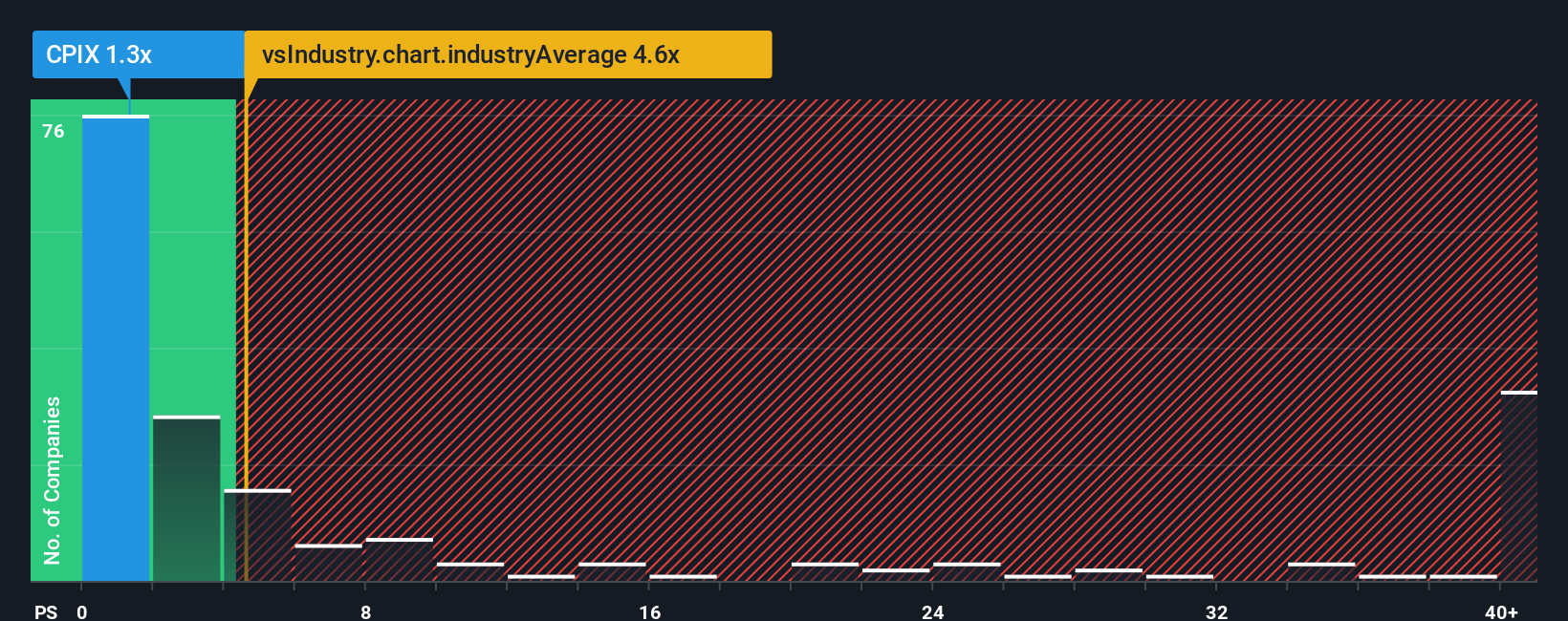
2026-02-06 11:58:33
Cumberland Pharmaceuticals Inc.'s shares have dropped 31% in the last month and 20% in the past year. Despite a 1.3x Price-to-Sales (P/S) ratio making it seem undervalued compared to the industry average of 4.6x, analysts believe the low P/S is justified due to inconsistent revenue growth. The company's recent 12% revenue gain lags the industry's predicted 23% growth, suggesting investors anticipate continued limited growth and are unwilling to pay a higher premium for the stock.

2026-02-06 10:58:33
Cumberland Pharmaceuticals (NASDAQ:CPIX) saw its stock price surpass its 200-day moving average, trading as high as $3.86 and closing at $3.59. Despite this technical performance, analyst sentiment for the specialty pharmaceutical company remains largely negative, with an average "Sell" rating due to its small market capitalization, quarterly loss, negative net margin, and relatively low institutional ownership. The article questions whether this rise signifies a selling opportunity given the underlying fundamental concerns and analyst outlook.

2026-02-05 04:28:43
Cumberland Pharmaceuticals Inc. (NASDAQ:CPIX) has received Fast Track Designation from the FDA for its oral therapy, ifetroban, aimed at treating heart disease in patients with Duchenne muscular dystrophy (DMD). This designation will accelerate the development and review process for the drug, which previously showed promising Phase 2 trial results with a 5.4% improvement in left ventricular ejection fraction. Ifetroban previously received Orphan Drug Designation and Rare Pediatric Disease Designation, highlighting the critical unmet medical need in DMD-related heart disease.
2026-02-04 22:28:43
Cumberland Pharmaceuticals Inc. (CPIX) announced on February 4, 2026, that its heart disease therapy for Duchenne muscular dystrophy (DMD), Ifetroban, has received FDA Fast Track Designation. This designation aims to accelerate the development and review of life-saving therapies and follows previous Orphan and Rare Pediatric Disease statuses given to the drug. A Phase 2 trial of Ifetroban showed a 5.4% improvement in heart function for DMD patients.


