
Adial Pharmaceuticals Inc
$ 2.53
3.69%
26 Feb - close price
- Market Cap 2,711,300 USD
- Current Price $ 2.53
- High / Low $ 2.66 / 2.35
- Stock P/E N/A
- Book Value 4.72
- EPS -15.00
- Next Earning Report 2026-03-03
- Dividend Per Share N/A
- Dividend Yield 0 %
- Next Dividend Date -
- ROA -0.79 %
- ROE -1.55 %
- 52 Week High 21.38
- 52 Week Low 2.27
About
Adial Pharmaceuticals, Inc., a clinical-stage biopharmaceutical company, focuses on the development of therapies for the treatment or prevention of addictions and related disorders. The company is headquartered in Charlottesville, Virginia.
Analyst Target Price
$26.00
Quarterly Earnings
| Sep 2025 | Jun 2025 | Mar 2025 | Dec 2024 | Sep 2024 | Jun 2024 | Mar 2024 | Dec 2023 | Sep 2023 | Jun 2023 | Mar 2023 | Dec 2022 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reported Date | 2025-11-13 | 2025-08-13 | 2025-05-12 | 2025-03-31 | 2024-11-12 | 2024-08-13 | 2024-05-14 | 2024-02-20 | 2023-11-14 | 2023-08-21 | 2023-05-12 | 2023-03-30 |
| Reported EPS | -0.08 | -0.18 | -0.34 | -0.15 | -0.38 | -0.59 | -0.62 | -1.35 | -1.18 | 0.96 | -0.11 | -0.105 |
| Estimated EPS | -0.39 | -0.39 | -0.495 | -0.33 | -0.44 | -0.53 | -0.33 | -0.76 | -1.13 | -1.5 | -0.14 | -0.15 |
| Surprise | 0.31 | 0.21 | 0.155 | 0.18 | 0.06 | -0.06 | -0.29 | -0.59 | -0.05 | 2.46 | 0.03 | 0.045 |
| Surprise Percentage | 79.4872% | 53.8462% | 31.3131% | 54.5455% | 13.6364% | -11.3208% | -87.8788% | -77.6316% | -4.4248% | 164% | 21.4286% | 30% |
Next Quarterly Earnings
| Dec 2025 | |
|---|---|
| Reported Date | 2026-03-03 |
| Fiscal Date Ending | 2025-12-31 |
| Estimated EPS | -2.1 |
| Currency | USD |
Next Dividend Records
| Dividend per share (year): | - |
| Dividend Yield | - |
| Next Dividend Date | - |
| Ex-Dividend Date | - |
Recent News: ADIL
2026-02-25 09:54:05
Adial Pharmaceuticals has successfully regained full compliance with the Nasdaq listing requirement. This announcement was made via Reuters and indicates the company's adherence to the stock exchange's standards.
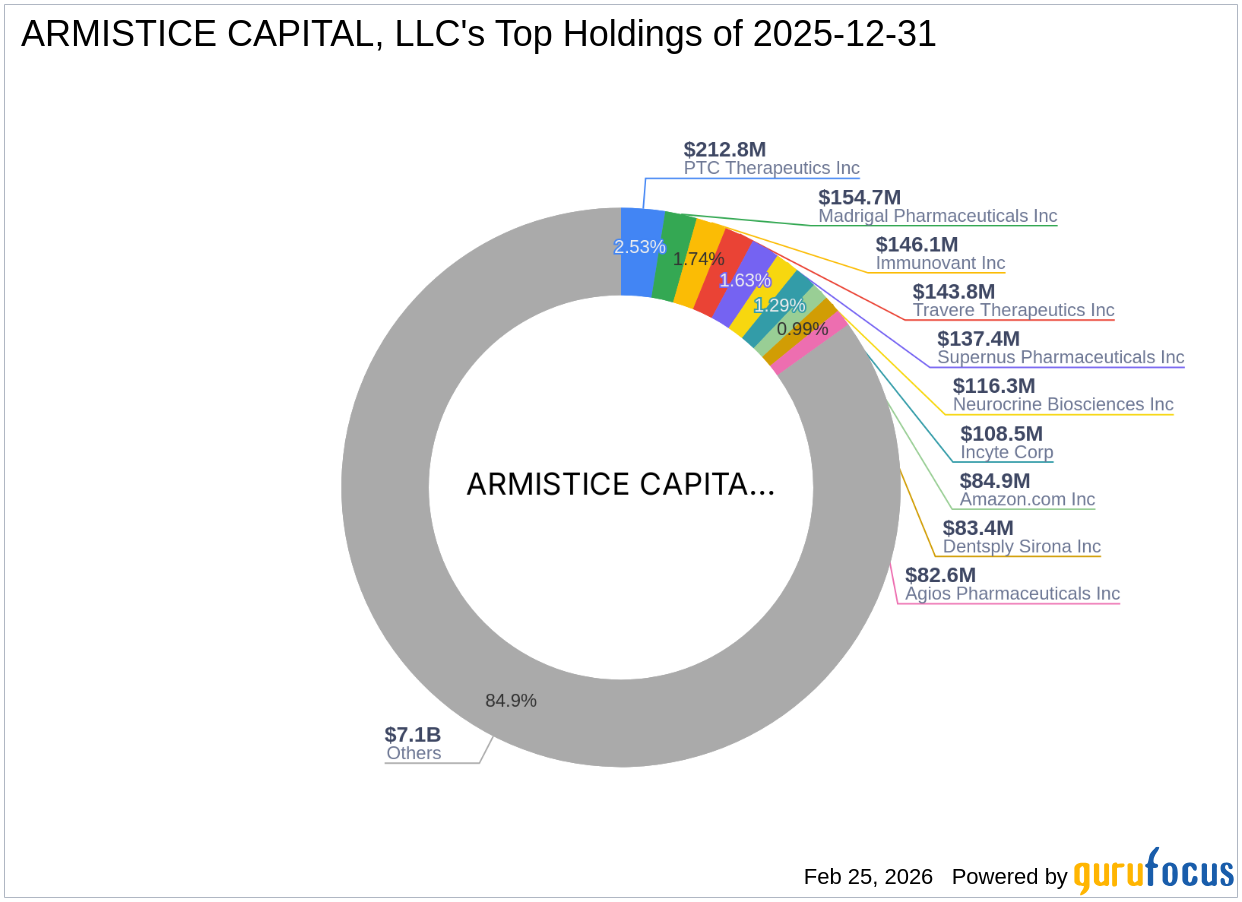
2026-02-25 00:53:09
ARMISTICE CAPITAL, LLC acquired 111,985.72 shares of Adial Pharmaceuticals Inc on December 31, 2025, marking a new holding that represents 9.99% of the company's outstanding shares. Despite Adial Pharmaceuticals' current financial challenges and significant stock price decline, Armistice Capital, known for its focus on healthcare and technology, likely sees long-term potential in Adial's innovative addiction treatment, AD04. This strategic investment underscores Armistice Capital's commitment to capitalizing on opportunities within the healthcare sector.
2026-02-24 19:53:09
Adial Pharmaceuticals (ADIL) has successfully met Nasdaq's minimum bid price requirement, with its stock closing above $1.00 for ten consecutive business days. The company is now progressing its lead product, AD04, into a crucial Phase 3 trial and is actively pursuing strategic partnerships to support its development.

2026-02-24 14:53:09
Adial Pharmaceuticals announced it has regained compliance with Nasdaq's minimum bid price rule, resolving a significant listing risk. The company is also moving forward with a new Phase 3 program for its lead candidate AD04, a genetically targeted therapy for Alcohol Use Disorder, and exploring its potential for other addiction indications. This progress comes as Adial indicates advanced strategic partnering discussions for AD04's development and commercialization.

2026-02-24 13:53:09
Adial Pharmaceuticals, Inc. announced it has regained full compliance with Nasdaq's minimum bid price requirement. The company confirmed its common stock maintained a closing bid price of $1.00 or greater for 10 consecutive business days, resolving the compliance issue. Adial is now focusing on advancing its AD04 therapeutic toward a pivotal Phase 3 program and is engaged in strategic partnering discussions for its development and commercialization.

2026-02-23 19:20:14
Adial Pharmaceuticals (ADIL) shares rose following an FDA policy shift that could reduce the number of pivotal trials required for drug approval. This new guidance allows for drug approval with one adequate clinical investigation plus confirmatory evidence, potentially lowering Phase 3 costs and accelerating the New Drug Application process for their lead candidate, AD04, which treats Alcohol Use Disorder. The company plans to engage with the FDA to align its AD04 strategy with these updated regulatory standards.


